Green Chemistry and Nano chemistry
Maharashtra Board-Class-12-Chemistry-Chapter-16
Notes
|
Topics to be Learn :
|
Introduction
- The concept of green chemistry was coined by Paul T. Anastas.
- Green Chemistry is the application of chemistry to decrease or eliminate the usage or creation of hazardous compounds by environmentally conscious design of chemical products and processes.
Sustainable development :
- Sustainable development is the development that meets the needs of the present, without compromising the ability of future generations to meet their own needs.
- We can attain sustainable development by using the twelve green chemistry principles.
Principles of green chemistry :
(i) Prevention of waste or by products :
- To prevent generating waste, there is the need to develop the zero waste technology (ZWT).
- ZWT in a chemical synthesis should result in waste product being zero or minimum.
- To use the waste product of one system as the raw material for other system is also the aim of ZWT.
Example :
- Cement and brick industry can use the bottom ash of thermal power station as the raw material.
- Effluent coming out from cleansing of machinery parts may be used as coolant water in thermal power station.
(ii) Atom economy :
Atom economy is a measure of the amount of atoms from the starting material that are present in the final product at the end of a chemical process.
Good atom economy means most of the atoms of the reactants are incorporated in the desired products. Only small amount of waste is produced, hence lesser problem of waste disposal.
The atom economy of a process can be calculated using the following formula.

Example :
(a) conversion of Butan-1-ol to 1 - bromobutane
CH3CH2CH2CH2OH + NaBr + H2SO4 → CH3CH2CH22CH2-Br + NaHSO4 + H2O
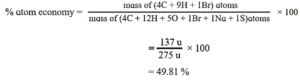
(b) Atom economy of fermentation of sugar (glucose) to ethanol. (2 marks)
C6H12O6(aq) → 2C2H5OH(aq) +2CO2(g)
Atomic masses of C = 12, H = 1, O = 16

Formula wt of glucose = 180
Formula wt of ethanol = 46
Relative mass/wt of desired useful product in the equation = 2 x 46 = 92
% Atom economy = 92/180 x 100 = 51.1%
(iii) Less hazardous chemical synthesis :
- The safest possible design for chemical reactions and synthesis processes is necessary to prevent the production of hazardous waste during chemical operations.
- It was discovered that the pesticide DDT (diphenyltrichloroethane), which was once in use, was hazardous to humans. As a result, benzene hexachloride (BHC) has taken the position of DDT as an insecticide, one of the γ-isomer (gamma) of BHC is called gammexane or lindane.
(iv) Designing Safer Chemicals :
This principle is quite similar to the previous one.
- There is a need to develop safer chemicals to protect chemical workers from exposure to harmful environments.
- Adipic acid is extensively used in polymer industry. In synthesis of adipic acid, benzene is used as the starting material, but benzene is carcinogenic and being volatile organic compound (VOC) it pollutes the air and environment.
- To overcome this health hazard Green technology developed by Drath and Frost, adipic acid is enzymatically synthesised from glucose.
(v) Use Safer solvent and auxiliaries :
The main aim behind this principle is to use green solvents.
- It states that safer solvent like water, supercritical CO, should be used in place of volatile halogenated organic solvents, like CH2Cl2, CHCl2, CCl4 for chemical synthesis and other purposes.
- Solvents dissolve solutes and form solutions, they facilitate many reactions. Water is a safer benign solvent while solvents like dichloromethane (CH2Cl2), chloroform (CHCl3) etc. are hazardous. Use of toxic solvents affect millions of workers every year and have implications for consumers and the environment. A large amount of waste is created by their use and they also have huge environmental and health impacts.
- Finding safer solvents or designing processes which are solvent free is the best way to improve the process and the product.
- Choose the safer solvent available for any given step of reaction. Minimize the total amount of solvents and auxilary substances used, as these make up a large percentage of the total waste created.
(vi) Design for energy efficiency :
- The amount of energy required for chemical synthesis is enormous. It is preferable to carry out reactions at room temperature and pressure in order to save energy.
- This may be accomplished by employing green chemistry principles such as the utilization of catalysts, microorganisms or biocatalysts, and renewable materials, among others.
- Less energy may be used by upgrading heating system technology, using microwaves, and so on.
(vii) Use of renewable feedstocks :
The concept is mostly concerned with petrochemicals.
- Industries use a lot of non-renewable feed stocks like petroleum. These resources are depleting fast and the future generation will be deprived. The excessive use of these resources have also put a burden on the environment.
- If renewable resources like agricultural or biological products are used, this will ensure the sharing of resources by future generations. This practice will also not put a burden on the environment.
- The products and waste produced are generally biodegradable and environmental friendly hence leading to a sustainable future.
(viii) Reduce derivatives : [Minimization of steps]
- A commonly used technique in organic synthesis is the use of protecting or blocking group.
- Finally, on completion of reaction deprotection of the group is required. This leads to unnecessary increase in the number of steps and decreased atom economy.
- The green chemistry principle aims to develop processes to avoid necessary steps i.e. (minimization of steps).
- When biocatalyst is used very often there is no need for protection of selective group.
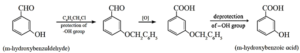
Example : Conversion of m-hydroxyl benzaldehyde to m-hydroxybenzoic acid.
| Know This :
Plastic packaging impact on the food :
|
(ix) Use of catalysis :
- A catalyst is a material that accelerates the speed of a reaction without being chemically altered in the process.
- Catalyst helps to increase selectivity, minimize waste and reduce reaction times and energy demands.
Examples :
- Hydrogenation of oil the catalyst used are platinum or palladium, Raney nickel.
- Haber’s process of manufacture of ammonia : catalyst used - Iron
- Manufacture of HDPE polymer : catalyst used - Zeigler-Natta catalyst
- Manufacture of H2SO4 by contact process : catalyst used - Vanadium oxide (V2O5)
- Fischer-Tropsch process (synthesis of gasoline) : catalyst used - Cobalt based or Iron based
(x) Design for degradation :
- Environment protection is the prime concern which has lead to the need for designing chemicals that degrade and can be discarded easily. These chemicals and their degradation products should be non-toxic, non-bioaccumulative or should not be environmentally persistent.
- The aim behind this principle is that the waste product should degrade automatically to clean the environment. Thus the preference for biodegradable polymers and pesticides.
- To make the separation and segregation easier for the consumer an international plastic recycle mark is printed on larger items.
(xi) Real-time Analysis Pollution Prevention:
- Analytical methods need to be further developed to allow for real-time, in process monitoring and control prior to the formation of hazardous substances.
- It is very much important for the chemical industries and nuclear reactors to develop or modify analytical methodologies so that continuous monitoring of the manufacturing and processing unit is possible.
(xii) Safer chemistry for Accident prevention:
We need to develop chemical processes that are safer and minimize the risk of accidents.
- It is important to select chemical substances used in a chemical reaction in such a way that they can minimize the occurrence of chemical accidents, explosions, fire and emissions.
- Example : Chemical process that works with the gaseous substances can lead to relatively higher possibilities of accidents including explosion as compared to the system working with nonvolatile liquid and solid substances.
The role of Green chemistry :
The green chemistry approach recognizes that the Earth does have a natural capacity for dealing with much of the waste and pollution that society generates.
When the waste and pollution that society generates exceeds the Earth’s natural capacity for dealing with it, the green chemistry approach plays an important role.
- Green chemistry reduces or eliminates the use or creation of hazardous compounds in the design, manufacture, and usage of chemical goods by supporting novel chemical technologies.
- Green chemistry reduces capital investment necessary for pollution protection. Green chemistry benefits industrial ecology because it includes and encourages pollution avoidance methods in the chemical production process.
- Green chemistry helps to protect the presence of ozone in the stratosphere. Ozone layer is essential for the survival of life on the earth.
- Global warming (Greenhouse effect) is controlled by green chemistry.
- At present it is the beginning of the green revolution. It is an exciting time with the new challenges for chemist involved with the discovery, manufacturing and use of chemicals.
- Green chemistry helps us to save environment and save earth, which is important for our future.
Introduction to nano chemistry :
- Nanochemistry : It is the combination of chemistry and nanoscience. It deals with designing and synthesis of materials of nanoscale with different size and shape, structure and composition and their organization into functional architectures.
- Nanoscience : The study of phenomena and manipulation of materials at atomic, molecular and macromolecular scales where properties differ significantly from those at a larger scale is called nanoscience.
- Nanotechnology : The design, characterization, production and application of structures, device and system by controlling shape and size at nanometer scale is called nanotechnology.
- Nanomaterial : A material having structural components with at least one dimension in the nanometer scale that is 1 - 100 nm is called the nanomaterial. Nanomaterials are larger than single atoms but smaller than bacteria and cells.
Classification of nanomaterials : Nanoparticles, nanowires and nanotubes can be classified according to dimensions. The nano structured materials may be large organic molecules, inorganic cluster compounds and metallic or semiconductor particles.
Zero, one and two dimensional nanoscale system :
- Zero-Dimensional Nanostructures : A zero-dimensional structure is one in which all three dimensions are in the nanoscale. For example : Nanoparticles.
- One-Dimensional Nanostructures : A one-dimensional nanostructure is one in which two dimensions are in the nanoscale. For example : Nanowires and Nano rods.
- Two-Dimensional Nanostructures : A two-dimensional nanostructure is one in which one dimension is in the nanoscale. For example : Thin films.
| Know This :
A very highly useful application of nanochemistry is ‘medicine’. A simple skin care product of nanochemistry is sunscreen. Sunscreen contains nanoparticles of Zinc oxide, (ZnO) and Titanium dioxide, (TiO2). These chemicals protect the skin against harmful UV (ultraviolet) rays by absorbing or reflecting the light and prevent the skin from damage. |
Characteristic features of Nanoparticles
The nanoparticle science is special as at such a small scale, different laws dominate than what we experience in our everyday life.
The characteristic features like optical properties, catalytical activities, have huge surface area and good thermal properties mechanical strength electrical conductivity vary than that of bulk material.
Colour :
- It is an optical property that is different at nanoscale.
- Example : Elemental gold has nice shining yellow colour, but nanoparticles of gold show red colour.
Surface area :
- High surface-to-volume ratio is a very important characteristic of nanoparticles. Bulk material if subdivided into a group of individual nanoparticles, the total volume remains the same, but the collective surface area is largely increased.
- With large surface area for the same volume, these small particles react much faster because more surface area provides more number of reaction sites, leading to more chemical reactivity.
- Explanation of increase in surface area with decrease in particle size.

Catalytic activity :
- Since the surface area of nanoparticles is large they show increased catalytic activity. They are usually heterogenous catalyst that means catalysts are solid form and the reactions occur on the surface of the catalyst. These catalysts can be easily separated and recycled.
- For example : Pd, Pt metal nanoparticles used in hydrogenation reactions. TiO,, ZnO are used in photocatalysis. Gold in bulk is unreactive but the nanoparticles of gold behave as very good catalyst for organic reactions.
Thermal properties : melting point :
- The melting point of nanomaterial changes drastically with size.
- For example : Sodium clusters (Nan) of 1000 atoms melts at 288 K, 10000 atoms melt at 303 K and bulk sodium melts at 371 K.
Mechanical properties :
- The mechanic al strength of nano clusters increase the hardness of the metal.
- For example : nanoparticles of copper and palladium clusters with diameter in the range of 5-7 nm have hardness up to 500 r. greater than the bulk metal.
Electrical conductivity :
- Electrical conductivity is observed to change at nanoscale.
- For example, carbon nanotube can act as a conductor or semiconductor in behaviour.
Synthesis of nanomaterials
There are two approaches to the synthesis of nanomaterials. (i) Bottom up and (ii) Top down

(i) Bottom-up method : Synthesis of nanoparticles in the bottom-up approach molecular components arrange themselves into more complex assemblies atom by atom, molecule by molecule and cluster by cluster from the bottom.
- Example : synthesis of nanoparticles by colloidal dispersion.
(ii) Top-down method : In the top-down approach, involves nano-materials being synthesised from bulk material by breaking the material. The bulk solids are disassembled into finer pieces until they are constituted of only few atoms.
- Example : Nanoparticles are synthesised by colloidal dispersion.
Wet chemical synthesis of Nanomaterials :
- Sol : Sols are dispersions of colloidal particles in a liquid. Colloids are solid particles with diameter of 1-100 nm.
- Gel : A gel is interconnected rigid network with pores of submicrometer dimensions and polymeric chains whose average length is greater than a micrometer.
Sol-gel Process : A sol-gel process is an inorganic polymerisation reaction. It is generally carried out at room temperature. It includes four steps : Hydrolysis, polycondensation, drying and thermal decomposition. This method is widely used to prepare oxide materials.
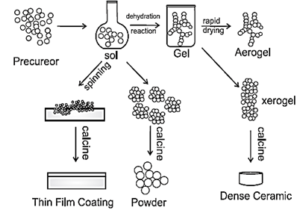
The rections involved in the sol-gel process can be described as follows :
MOR + H2O → MOH + ROH ..(hydrolysis)
metal alkoxide
MOH + ROM → M-O-M + ROH ..(condensation)
- Formation of different stable solution of the alkoxide or solvated metal precursor
- Gelation resulting from the formation of an oxide or alcohol-bridged network. (gel) by a polycondensation reaction.
- Aging of the gel means during that period gel transforms into a solid mass.
- Drying of the gel : In this step, water and other volatile liquids are removed from the gel network.
- Dehydration : The material is heated at temperatures upto 800 0
Analysis or characterization of nanomaterials :
The synthesized material is analyzed by various analytical tools or techniques. The analytical tools used for characterization of nanoparticles are
- V visible spectroscopy : It gives the preliminary confirmation of formation of nanoparticles.
- X-ray Diffraction (XRD) : The information given by this tool is about particle size, crystal structure and geometry.
- Scanning electron microscopy (SEM) : This is used to study the structure of surface of material that is the morphology of the material.
- Transmission electron microscopy (TEM) : It gives information about the particles size.
- Fourier transform infrared spectroscopy (FTIR) : It gives information about absorption of functional groups and binding nature of the nanomaterial.
History of nanotechnology :
There is enough evidence that nanomaterials have been produced and used by humans for hundreds of years.
Examples :
- Gold and silver nanoparticles trapped in the glass matrix gives ruby red colour in some ancient glass paintings.
- The decorative glaze or metallic film known as lustre found on some medieval pottery is due to certain spherical metallic nanoparticles.
- Carbon black is a nanostructured material that is used in tyres of car to increase the life of tyre. (Discovery in 1900). Carbon nanotubes are made up of graphite sheets with nanosized diameter. They have highest strength.
- Fumed silica, a component of silicon rubber, coatings, sealants and adhesives is also a nanostructured material.
Applications of nanomaterials :
The contribution of nanochemistry in number of innovative products in various disciplines due to their unique physical, chemical, optical, structural, catalytic properties. Few applications are as follows :
- Nanoparticles contribute to stronger, lighter, cleaner and smarter surfaces and systems. They are used in the manufacture of scratchproof eyeglasses, transport, sunscreen, crack resistant paints, etc.
- Used in electronic devices like Magnetoresistive Random Access Memory (MRAM).
- Nanotechnology plays an important role in water purification techniques. Silver nanoparticles act as highly effective bacterial disinfectant, remove E.Coli from water. They are used in water purification system to get safe drinking water.
- Self cleansing materials : Lotus is an example of self cleansing. Nanostructures on lotus leaves repel water which carries dirt as it rolls off. Lotus effect is the basis of self cleaning windows.
Nanoparticles and Nanotechnology :
Advantages :
- Revolution in electronics and computing.
- Energy sector - nanotechnology will make solar power more economical. Energy storage devices will become more efficient.
- Medical field : Manufacturing of smart drugs, helps cure faster and without side effects. Curing of life threatening diseases like cancer and diabetes.
Disadvantages :
Despite the benefits that nanotechnology offers to the world, it is accompanied by certain disadvantages and potential risks.
- The standard of living has been raised by nanotechnology but at the same time it has increased the environmental pollution. The kind of pollution caused by nanotechnology is very dangerous for living organism, it is called nano pollution.
- Nanoparticles can be potential health hazard depending on the size, chemical composition and shape.
- They can be inhaled and can be deposited in the human respiratory tract and in the lungs, causing lung damage.
PDF : Class-12-Chemistry-Chapter-16-Green Chemistry and Nano chemistry-Text Book
PDF : Class-12-Chemistry-Chapter-16-Green Chemistry and Nano chemistry- Notes
PDF : Class-12-Chemistry-Chapter-16-Green Chemistry and Nano chemistry- Solution
All 16 Chapters Notes -Class-12-Chemistry (16-PDF)
All 16 Chapters Solutions -Class-12-Chemistry (16-PDF)
All 16 Chapters Notes+Solutions -Class-12-Chemistry (32-PDF)
Main Page : – Maharashtra Board Class 12th-Chemistry – All chapters notes, solutions, videos, test, pdf.
Previous Chapter : Chapter 15- Introduction to Polymer Chemistry – Online Notes
We reply to valid query.