Heat Transfer in Nature
NCERT-Class-7-Science (Curiosity)-Chapter-7-CBSE
Notes
|
Topics to be Learn :
|
Introduction :
- Heat is a type of energy that makes things feel hot or cold.
- We cannot see heat, but we can feel it.
- In winter, we feel cold inside the house, but when we go into sunlight, we feel warm. In summer, we feel hot even inside the house.
- This shows that how hot or cold something feels depends on the amount of heat in it.
- Temperature helps us measure how hot or cold something is.
Relative Nature of Heat :
In our daily routine, we come across a number of objects, out of which some objects are hot while other objects are cold. e.g.
- A frying pan kept on a burning gas stove becomes hot but the handle of pan is cold.
- When we touch a cup of freshly made tea, it feels hot but when we touch boiling water, it feels hotter.
Hence, the hotness and coldness of an object are relative.
- The feeling of hotness or coldness is not always the same for everyone.
- It depends on comparison with other objects or surroundings.
- The best way to know how hot or cold something is, is by measuring its temperature.
Transfer of Heat :
Heat moves from a hot object to a cold object. In other words, heat flows from a higher temperature to a lower temperature. This movement of heat is called transfer of heat.
Example : If we put a steel spoon in a cup of hot tea, the spoon becomes hot. This happens because heat from the tea moves into the spoon.
When Does Heat Stop Flowing?
- Heat keeps flowing until both objects have the same temperature.
- When they reach the same temperature, heat stops flowing.
Ways of Heat Transfer : There are three ways in which heat can transfer:
(i) Conduction : Heat transfer in solids.
(ii) Convection : Heat transfer in liquids and gases.
(iii) Radiation : Heat transfer through empty space (vacuum), without any medium.
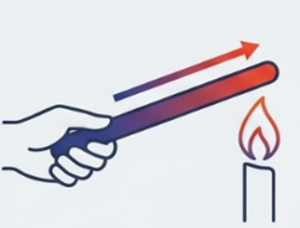
Conduction of Heat :
Conduction : Conduction is the process of heat transfer from a hotter part to a colder part through direct contact.
- In this process, the object itself does not move.
- The particles stay in their place but pass heat to nearby particles.
- It mainly occurs in solids.
Example : When an iron pin is heated at one end, the other end also becomes hot.
Rate of Heat Transfer : Heat flows faster when the temperature difference between objects is more.
Conductors and Insulators of Heat :
Materials react differently to heat based on their molecular structure.
| Category | Definition | Examples |
| Conductors | Materials that allow heat to pass through them easily. | Metals (Iron, copper, silver, aluminum). |
| Insulators | Materials that do not allow heat to pass through easily; also called poor conductors. | Glass, plastic, wood, water, air, clay, porcelain. |
Practical Applications :
- Cookware: Utensils are made of metal (conductors) to transfer heat to food, while handles are made of plastic or wood (insulators) for safe handling.
- Clothing:
- Dark-colored objects absorb and emit heat better than light-colored objects, making them suitable for winter.
- Light-colored objects reflect heat, making them ideal for summer.
- Woolen clothes keep us warm because wool is a poor conductor and traps air, which further prevents body heat from escaping.
- Construction: Hollow bricks are used in hot regions because the trapped air acts as an insulator, keeping the interior of the house cool.
Convection :
- Convection is the process of heat transfer in liquids and gases.
- In this process, heat is transferred by the actual movement of particles.
- Convection happens only in liquids and gases, not in solids.
How Does It Happen? :
- When a liquid or gas is heated, the particles become lighter and move upward.
- Cooler and heavier particles move downward to take their place.
- This movement creates a cycle called a convection current, which transfers heat.
Examples of Convection :
- Boiling water: Water at the bottom gets heated, moves up, and cooler water comes down.
- Sea breeze: During the day, cool air from the sea moves towards the land.
- Room heater: Warm air rises and spreads in the room, while cool air moves down to get heated.
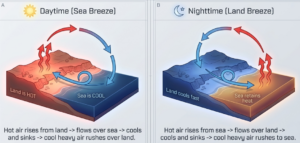
Land and Sea Breeze :
Sea breezes and land breezes are natural phenomena that occur due to convection, which is the transfer of heat through the actual movement of particles in liquids and gases. These breezes are caused by the fact that land and water heat up and cool down at different rates.
Sea Breeze (During the Day)
- Heating: During the day, the Sun heats the land much faster than the water in the sea.
- Rising Air: The air above the land becomes warmer and lighter, causing it to rise.
- Convection Flow: As this warm air rises, cooler and heavier air from the sea moves in toward the land to take its place. This movement of cooler air from the sea to the land is called a sea breeze.
Land Breeze (At Night) :
- Cooling: At night, the process reverses because the land cools down much faster than the sea water.
- Rising Air: Consequently, the air above the sea is now warmer than the air above the land, causing the warmer air over the sea to rise.
- Convection Flow: Cooler air from the land moves toward the sea to replace the rising warm air, creating what is known as a land breeze.
In both cases, these winds are driven by convection currents, where the movement of air particles transfers heat and balances temperature differences between the land and the sea.
Radiation :
Radiation is the transfer of heat that does not require a material medium. Heat travels directly from a source (like the Sun or a fireplace) to an object.
- All objects radiate heat to their surroundings.
- Can occur in a vacuum or free space.
- Heat travels via rays (infrared waves).
Example: Solar energy reaching Earth from space or feeling the warmth of a fireplace from a distance.
Kinds of Clothes we Wear in Summer and Winter :
The types of clothes we wear in different seasons are determined by the principles of heat transfer and insulation.
Summer Clothing : During the summer, it is more comfortable to wear white or light-colored clothes.
- Why: Light-colored clothes reflect most of the heat that falls on them. By reflecting this heat rather than absorbing it, these clothes help keep the body cooler and more comfortable in hot weather.
Winter Clothing : In the winter, we typically wear woollen clothes and dark-colored clothes to stay warm.
- Woollen Clothes: Wool is effective because it traps air within its pores. Since air is a poor conductor of heat, it acts as an insulator that reduces the flow of heat from our bodies to the colder surroundings, helping us retain body warmth.
- Dark-Colored Clothes: Dark surfaces absorb more heat from the surroundings, such as sunlight, making them more comfortable to wear when it is cold.
- Layering: Wearing multiple layers of clothing is often warmer than wearing one thick garment. This is because air is trapped between the layers, and since trapped air is a poor conductor, it provides additional insulation against heat loss.
Architecture and Insulation :
- Himalayan Construction: In regions like Uttarakhand, houses use double wooden walls filled with cow dung and mud. Because wood and mud are poor conductors, they prevent heat loss during extreme winters.
- Hollow Bricks: Outer walls in hot or cold regions often use hollow bricks. The trapped air inside acts as an insulator, keeping the interior warm in winter and cool in summer.
- The Bukhari: A traditional Himalayan room heater that utilizes all three heat transfer methods:
- Conduction: The flat metal top transfers heat to cooking utensils.
- Convection: The chimney vents smoke, and hot air circulates through the room.
- Radiation: Thermal energy is emitted directly from the iron stove to the occupants.
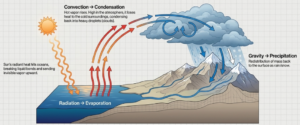
Water Cycle :
- The water cycle is the continuous movement of water on Earth.
- Water moves up into the air as water vapour and comes back as rain, snow, or hail.
- It helps to supply water to rivers, lakes, and oceans.
- The total amount of water on Earth remains the same.
Stages of the Water Cycle :
(i) Evaporation and Transpiration :
- The Sun gives heat energy to the Earth. This heat changes water into water vapour.
- Evaporation: Water from oceans, rivers, and lakes turns into vapour.
- Transpiration: Plants release water vapour from their leaves.
(ii) Condensation and Precipitation :
- Condensation: Water vapour rises, cools, and forms clouds.
- Precipitation: When clouds become heavy, water falls as rain, snow, or hail.
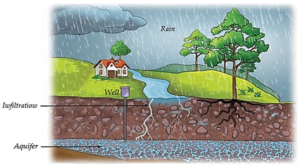
(iii) Infiltration and Groundwater :
- Infiltration: Some water seeps into the soil.
- This water is stored underground as groundwater.
- Underground water storage areas are called aquifers.
Water from the ground and surface flows back into rivers and oceans. Then the cycle starts again.
States of Water in the Cycle :
- Liquid: Found in rivers, lakes, and oceans.
- Solid: Found as snow, ice, and glaciers.
- Gas: Found as water vapour in the air.
How Water Cycle Helps in Heat Distribution :
- Absorption of Heat : Water absorbs heat from the Sun and evaporates.
- Heat Transfer by Convection : Water vapour rises and carries heat into the atmosphere.
- Release of Heat : When vapour cools and forms clouds, heat is released. This helps control weather.
- Temperature Control : Large water bodies heat up slowly and cool down slowly. This keeps nearby areas from becoming too hot or too cold.
The water cycle moves water between land, air, and oceans. It also helps in spreading heat across the Earth. It is very important for life and weather on Earth.
Seepage of water beneath the Earth :
Seepage is the process by which water on the Earth's surface, such as rainwater, sinks into the ground. This process is scientifically known as infiltration.
How Water Seeps Through the Surface :
- Water moves downward by passing through the spaces between soil and rock particles.
- The rate at which water seeps depends on the type of material it is passing through. Based on experiments:
- Gravel: Water seeps fastest because the spaces between particles are wider.
- Sand: Seepage is slower than in gravel.
- Clay: Water seeps slowest because the particles are very fine with minimal space between them.
- Infiltration happens most easily when the underground spaces are wide, open, and interconnected.
Storage and Groundwater :
- Groundwater: Once water seeps beneath the surface, it is stored in the pore spaces of sediments and openings in rocks.
- Aquifers: The specific underground layers of sediments and rocks that store this water are called aquifers. This is the water source used when people dig wells or drill bore wells.
Challenges to Seepage :
Groundwater is a finite resource currently facing depletion due to:
- In modern urban areas, seepage is often limited because of increased concrete surfaces and decreased vegetation, which prevents water from entering the ground.
Innovative Solutions:
- Rainwater Harvesting: Using recharge pits to replenish aquifers.
- Ice Stupas (Ladakh): In winter, water from mountain streams is piped and sprayed into the cold air. It freezes into cone-shaped structures that melt slowly during spring, providing a critical water source for summer farming when mountain streams run dry.
Historical Context of Weather Prediction :
Varahamihira, a sixth-century CE astronomer and mathematician from Ujjaini. In his work Brihatsamhita, he detailed methods for predicting seasonal rainfall by observing cloud formations, wind patterns, and celestial positions, demonstrating a long-standing human endeavor to understand environmental cycles.
Key Features of Kitabcd Exam Master :
|
Click on below links to get PDF from store
PDF : Chapter-7-Heat Transfer in Nature– Notes
PDF : Chapter-7-Heat Transfer in Nature– Exam Master (Solutions)
Main Page : NCERT-Class-7-Science (Curiosity) – All chapters notes, solutions, videos, test, pdf.
Next Chapter : Chapter-6- Adolescence: A Stage of Growth and Change – Online Notes
Next Chapter : Chapter-8- Measurement of Time and Motion – Online Notes


We reply to valid query.