Nature of Matter: Elements, Compounds, and Mixtures
NCERT-Class-8-Science (Curiosity)-Chapter-8
Solutions (Exercise + Intext)
NCERT Probe & Ponder Questions :
Question 1. Which of the entities in the picture below consist of matter, and which of them do not?
Entities that consist of matter are school building, students, trees, metal railing, football, school bag, books, water bottle, lunch box, and clothes of students etc. Entities that do not consist of matter - light, sound, heat etc.
Question 2. How can elements be combined to form a compound?
Examples: (i) Hydrogen + Oxygen → Water (H₂O) (ii) Sodium + Chlorine → Sodium Chloride (NaCl)
Question 3. How could the discovery of a compound that absorbs carbon dioxide from the air contribute to solving environmental challenges?
If scientists discover a compound that can absorb carbon dioxide (CO₂) from the air, it can help the environment in many ways.
Keep The Curiosity Alive :
Question 1. Consider the following reaction where two substances, A and B, combine to form a product C: A + B → C
Assume that A and B cannot be broken down into simpler substances by chemical reactions. Based on this information, which of the following statements is correct?
(i) A, B, and C are all compounds and only C has a fixed composition.
(ii) C is a compound, and A and B have a fixed composition.
(iii) A and B are compounds, and C has a fixed composition.
(iv) A and B are elements, C is a compound, and has a fixed composition.
(iv) A and B are elements, C is a compound, and has a fixed composition.
Question 2. Assertion: Air is a mixture.
Reason: A mixture is formed when two or more substances are mixed, without undergoing any chemical change.
(i) Both Assertion and Reason are true and Reason is the correct explanation for Assertion.
(ii) Both Assertion and Reason are true, but Reason is not the correct explanation for Assertion.
(iii) Assertion is true, but Reason is false.
(iv) Assertion is false, but Reason is true.
(i) Both Assertion and Reason are true and Reason is the correct explanation for assertion. Explanation : Air is indeed a mixture because it is composed of various gases like nitrogen, oxygen, and carbon dioxide, which are mixed together without any chemical reaction between them. The properties of these individual gases are retained within the air.
Question 3. Water, a compound, has different properties compared to those of the elements oxygen and hydrogen from which it is formed. Justify this statement.
Water is a compound, and it behaves very differently from the elements that make it. But when they combine together, they form water, which is a liquid that actually puts out fire. This shows that a compound has completely new properties that are not the same as the elements it is made of.
Question 4. In which of the following cases are all the examples correctly matched? Give reasons in support of your answers.
(i) Elements — water, nitrogen, iron, air.
(ii) Uniform mixtures— minerals, seawater, bronze, air.
(iii) Pure substances— carbon dioxide, iron, oxygen, sugar.
(iv) Non-uniform mixtures — air, sand, brass, muddy water.
(i) Elements — water, nitrogen, iron, air. Incorrect Reason: So all examples are not elements. (ii) Uniform mixtures — minerals, seawater, bronze, air. Incorrect Reason: Because minerals are not mixtures, the list is incorrect. (iii) Pure substances — carbon dioxide, iron, oxygen, sugar. Correct Reason: All these are pure substances because each has only one type of particle: So all examples are correctly matched. (iv) Non-uniform mixtures — air, sand, brass, muddy water. Incorrect Reason: So this list is not correct. Final Answer: Only option (iii) is correctly matched.
Question 5. Iron reacts with moist air to form iron oxide, and magnesium burns in oxygen to form magnesium oxide. Classify all the substances involved in the above reactions as elements, compounds or mixtures, with justification.
Iron reacts with moist air to form iron oxide. Iron (Fe) + Oxygen (O2) + Water (H2O) → Iron Oxide (Fe2O3) Magnesium burns in oxygen to form magnesium oxide. Magnesium (Mg) + Oxygen (O2) → Magnesium Oxide (MgO) Classification of each substance as element, compound, or mixture. (1) Iron (Fe) → Element (2) Moist Air → Mixture (non-uniform or uniform depending on dust presence) (3) Iron Oxide (Fe₂O₃ / Fe₃O₄) → Compound (4) Magnesium (Mg) → Element (5) Oxygen (O₂) → Element (6) Magnesium Oxide (MgO) → Compound Reason: Formed by chemical reaction between magnesium and oxygen. Contains Mg and O in a fixed proportion. Final Classification Table :
Substance
Type
Reason
Iron
Element
Only Fe atoms
Moist Air
Mixture
Gases mixed physically
Iron Oxide
Compound
Fe and O chemically combined
Magnesium
Element
Only Mg atoms
Oxygen
Element
Only O atoms
Magnesium Oxide
Compound
Mg and O chemically combined
Question 6. Classify the following as elements, compounds, or mixtures in Table.
Carbon dioxide, sand, seawater, magnesium oxide, muddy water, aluminium, gold, oxygen, rust, iron sulfide, glucose, air, water, fruit juice, nitrogen, sodium chloride, sulfur, hydrogen, baking soda.
| Elements | Compounds | Mixtures |
Identify pure substances amongst these and list them below.
Pure substances :
Gold Oxygen Nitrogen Sulfur Hydrogen Magnesium oxide Rust (iron oxide) Iron sulfide (FeS) Glucose Water Sodium chloride (salt) Baking soda (sodium bicarbonate) Seawater Muddy water Air Fruit juice Pure Substances: Aluminium, gold, oxygen, nitrogen, sulfur, hydrogen, carbon dioxide, magnesium oxide, iron sulfide, glucose, water, sodium chloride, baking soda.
Elements
Compounds
Mixtures
Aluminium
Carbon dioxide
Sand
Question 7. What new substance is formed when a mixture of iron filings and sulfur powder is heated, and how is it different from the original mixture? Also, write the word equation for the reaction.
When a mixture of iron filings and sulfur powder is heated, a new substance called iron sulfide is formed. Difference from the original mixture : Word Equation : Iron + Sulfur → Iron sulfide
Original Mixture
After Heating
Just a mixture of iron and sulfur. Each substance keeps its own properties.
A compound called iron sulfide is formed.
Iron can be separated with a magnet; sulfur looks yellow.
Iron sulfide is a black compound that cannot be separated by physical methods.
No new substance is formed.
A new substance with new properties is formed.
Question 8. Is it possible for a substance to be classified as both an element and a compound? Explain why or why not.
No, a substance cannot be classified as both an element and a compound. Elements and compounds are two different, mutually exclusive categories: Element : Compound : A substance cannot be made of only one type of atom (element), and at the same time two or more different types of atoms chemically combined (compound). These definitions are opposite, so a single substance can fit only one category.
Question 9. How would our daily lives be changed if water were not a compound but a mixture of hydrogen and oxygen?
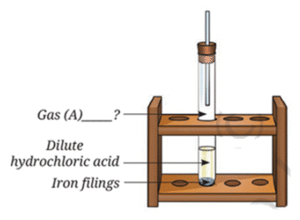
Question 10. Analyse Fig. Identify Gas A. Also, write the word equation of the chemical reaction.
By analysing the figure, it is found that there will be a chemical reaction inside the test tube between dilute HCl and Fe. 2HCl + Fe → FeCl2 + H2 ↑ So the reaction forms Iron Chloride (FeCl2) and the gas above will be Hydrogen (H2). Hydrochloric acid + Iron filing → Iron chloride + Hydrogen (g) Thus, Gas A = Hydorgen
Question 11. Write the names of any two compounds made only from non-metals, and also mention two uses of each of them.
(1) Carbon dioxide (CO₂) : Elements: Carbon + Oxygen (both non-metals) Uses: (2) Ammonia (NH₃) : Elements: Nitrogen + Hydrogen (both non-metals) Uses:
Question 12. How can gold be classified as both a mineral and a metal?
Gold can be classified as both a mineral and a metal because these two words describe different types of classifications. (1) Gold as a metal : Gold is a chemical element (symbol: Au). It has typical metallic properties, such as: So, based on physical and chemical properties, gold is a metal. (2) Gold as a mineral : A mineral is a naturally occurring, solid substance found in the Earth’s crust, with a definite composition. So, based on its natural occurrence in Earth, gold is a mineral. Therefore, gold fits both categories.
Key Features of Kitabcd Exam Master :
|
Click on below links to get PDF from store
PDF : Class 8 -Curiosity-Chapter-8-Nature of Matter: Elements, Compounds, and Mixtures– Notes
PDF : Class 8 -Curiosity-Chapter-8-Nature of Matter: Elements, Compounds, and Mixtures– Exam Master
PDF Set :
Class -8-Science (Curiosity) -All 13 Chapters Notes Set (13-PDF)-Rs.58
Class -8-Science (Curiosity) -All 13 Chapters Exam Master Set (13-PDF)-Rs.74
Class -8-Science (Curiosity) -All 13 Chapters Notes + Exam Master Set (26-PDF)-Rs.116
Main Page : NCERT-Class-8-Science (Curiosity) – All chapters notes, solutions, videos, test, pdf.
Previous Chapter : Chapter-7- Particulate Nature of Matter – Online Notes
Next Chapter : Chapter-9- The Amazing World of Solutes, Solvents, and Solutions – Online Notes

We reply to valid query.