Changes Around Us: Physical and Chemical
NCERT-Class-7-Science (Curiosity)-Chapter-4
Solutions (Exercise + Intext)
Intext Questions :
Question 1. Burning of magnesium is physical or chemical change? (Page 62)
Burning of magnesium is a chemical change.
Reason:
- When magnesium burns, it reacts with oxygen in the air
- A new substance called magnesium oxide is formed
- Heat and bright white light are also produced
Since a new substance is formed and the change cannot be reversed, burning of magnesium is a chemical change.
Question 2. Why does the candle that was covered with a glass tumbler stops burning after some time? (Page 63)
- A candle needs oxygen from the air to burn (combustion).
- When the candle is covered with a glass tumbler, fresh air cannot enter.
- The limited oxygen inside the tumbler gets used up after some time.
- Once the oxygen is finished, burning cannot continue, so the flame goes out.
Question 3. How can you test the presence of carbon dioxide gas? (Page 63)
Carbon Dioxide Test :
- The presence of CO2 is confirmed using lime water (calcium hydroxide).
- When CO2 is passed through lime water, it turns milky due to the formation of calcium carbonate, an insoluble white precipitate.
Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water
Question 4. Can physical and chemical changes occur in the same process? (Page 64)
Yes, both physical and chemical changes occur when a candle burns.
- Physical Change: The melting of wax, its solidification, and evaporation.
- Chemical Change: The burning of the wax vapor or the wick, which produces carbon dioxide and heat.
Question 5. Are changes permanent? (Page 66)
No, not all changes are permanent.
Some changes can be reversed like melting ice or boiling water, while others cannot be reversed, like chopping vegetables or making popcorn.
Question 6. Are all changes desirable? (Page 66)
No, not all changes are desirable.
Useful changes like making curd or cooking food are desirable, but
undesirable changes like the rusting of iron are not.
Question 7. How are sediments formed at the base of mountains? (Page 67)
Weathering is a slow natural process in which large rocks break down into smaller pieces called sediments.
These sediments collect at the base of mountains and help in the formation of soil.
Let Us Enhance Our Learning : Exercise Questions
Question 1. Which of the following statements are the characteristics of a physical change?
(i) The state of the substance may or may not change.
(ii) A substance with diff erent properties is formed.
(iii) No new substance is formed.
(iv) The substance undergoes a chemical reaction.
(a) (i) and (ii) (b) (ii) and (iii)
(c) (i) and (iii) (d) (iii) and (iv)
(c) (i) and (iii)
This is because in a physical change, the state of the substance may or may not change and no new substance is formed.
Question 2. Predict which of the following changes can be reversed and which cannot be reversed. If you are not sure, you may write that down. Why are you not sure about these?
(i) Stitching cloth to a shirt
(ii) Twisting of straight string
(iii) Making idlis from a batter
(iv) Dissolving sugar in water
(v) Drawing water from a well
(vi) Ripening of fruits
(vii) Boiling water in an open pan
(viii) Rolling up a mat
(ix) Grinding wheat grains to fl our
(x) Forming of soil from rocks
(i) Stitching cloth to a shirt : Irreversible - Once stitched, it cannot return to original cloth easily
(ii) Twisting of straight string : Reversible - Can be untwisted back to original form
(iii) Making idlis from batter : Irreversible - Cannot convert idli back to batter
(iv) Dissolving sugar in water : Reversible - Sugar can be obtained back by evaporation
(v) Drawing water from a well : Reversible - Water can be put back into the well
(vi) Ripening of fruits : Irreversible - Cannot change ripe fruit back to raw
(vii) Boiling water in an open pan : Reversible - Steam can be cooled back into water
(viii) Rolling up a mat : Reversible -Can be unrolled easily
(ix) Grinding wheat grains to flour : Irreversible - Cannot change flour back to grains
(x) Formation of soil from rocks : Irreversible - Cannot convert soil back into rocks easily
We may be not sure in some cases (like dissolving sugar or boiling water) because special methods are needed to reverse them.
Question 3. State whether the following statements are True or False. In
case a statement is False, write the correct statement.
(i) Melting of wax is necessary for burning a candle.
Statement is: True
Explanation:
- When a candle is lit, the wax near the flame melts.
- The melted wax is drawn up through the wick and burns.
- So, melting of wax is necessary for the candle to keep burning.
(ii) Collecting water vapour by condensing involves a chemical change.
Statement is: False
Correct Statement: Collecting water vapour by condensing involves a physical change.
Explanation:
- In condensation, water vapour changes into liquid water
- No new substance is formed
- Only the state changes (gas → liquid)
- So, it is a physical change, not a chemical change.
(iii) The process of converting leaves into compost is a chemical change.
Statement is: True
- Since a new substance is formed, it is a chemical change.
(iv) Mixing baking soda with lemon juice is a chemical change.
Statement is: True
- Since a new substance is formed and gas is produced, it is a chemical change.
Question 4. Fill in the blanks in the following statements:
(i) Nalini observed that the handle of her cycle has got brown deposits. The brown deposits are due to ________, and this is a________ change.
Nalini observed that the handle of her cycle has got brown deposits. The brown deposits are due to rusting, and this is a chemical change.
(ii) Folding a handkerchief is a _______ change and can be________.
Folding a handkerchief is a physical change and can be reversed.
(iii) A chemical process in which a substance reacts with oxygen with evolution of heat is called _________, and this is a __________ change.
A chemical process in which a substance reacts with oxygen with evolution of heat is called combustion, and this is a chemical change.
(iv) Magnesium, when burnt in air, produces a substance called __________. The substance formed is _________ in nature. Burning of magnesium is a _________ change.
Magnesium, when burnt in air, produces a substance called magnesium oxide. The substance formed is basic in nature. Burning of magnesium is a chemical change.
Question 5. Are the changes of water to ice and water to steam, physical or chemical? Explain.
Changes of Water to Ice and Water to Steam : Both changes are physical changes.
Explanation:
Water to ice (freezing):
- Liquid water changes into solid ice
- Only the state changes (liquid → solid)
- No new substance is formed
Water to steam (boiling):
- Liquid water changes into gas (steam)
- Only the state changes (liquid → gas)
- No new substance is formed
Since only the state changes and no new substance is formed, both changes are physical changes.
Question 6. Is curdling of milk a physical or chemical change? Justify your statement.
Curdling of milk is a chemical change.
Justification:
- Milk changes into curd, which is a new substance
- The properties of curd are different from milk (taste, texture, smell)
- The change cannot be reversed back to milk
Since a new substance is formed and the change is irreversible, curdling of milk is a chemical change.
Question 7. Natural factors, such as wind, rain, etc., help in the formation of soil from rocks. Is this change physical or chemical and why?
This change is actually both physical and chemical, but the dominant process depends on the factor:
Physical Changes :
- Wind: Breaks rocks into smaller fragments through abrasion.
- Rainwater (mechanical action): Causes cracks, erosion, and transportation of particles.
- These are physical changes because the rock’s composition remains the same, only its size and shape are altered.
Chemical Changes :
- Rainwater (chemical action): Slightly acidic rain (due to dissolved CO₂ forming carbonic acid) reacts with minerals in rocks.
- Oxidation: Oxygen in air reacts with minerals like iron, forming oxides (rust-like substances).
- These are chemical changes because new substances are formed with different properties.
Question 8. Read the following story titled ‘Eco-friendly Prithvi’, and tick the most appropriate option(s) given in the brackets. Provide a suitable title of your choice for the story.
Prithvi is preparing a meal in the kitchen. He chops vegetables, peels potatoes, and cuts fruits (physical changes/chemical changes). He collects the seeds, fruits, and vegetable peels into a clay pot (physical change / chemical change). The fruits, vegetable peels, and other materials begin to decompose due to the action of bacteria and fungi, forming compost (physical change / chemical change). He decides to plant seeds in the compost and water them regularly. After a few days, he notices that the seeds begin to germinate and small plants start to grow, eventually blooming into colourful flowers (physical change/chemical change). His eff orts are appreciated by all his family members.
(i) Chopping vegetables, peeling potatoes, and cutting fruits are physical changes.
(ii) Collecting seeds, fruits, and vegetable peels into a clay is a physical change.
(iii) Decomposition of fruits, vegetable peels, and other materials due to the action of bacteria and fungi to form compost is a chemical change.
(iv) Germination and growth of plants are chemical changes because new substances and structures are formed during growth.
The suitable title for the story is: "composting from kitchen waste".
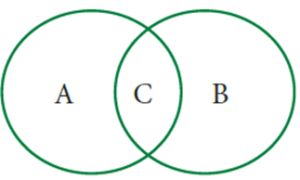
Question 9. Some changes are given here. Write physical changes in the area marked ‘A’ and chemical changes in the area marked ‘B’. Enter the changes which are both physical and chemical in the area marked ‘C’.
Process of burning a candle; Tearing of paper; Rusting; Curdling of milk; Ripening of fruits; Melting of ice; Folding of clothes; Burning of magnesium and Mixing baking soda with vinegar.
A : Tearing of paper, melting of ice, folding of clothes
B : Rusting, curdling of milk, ripening of fruits, burning of magnesium, mixing baking soda with vinegar
C : Burning of a candle
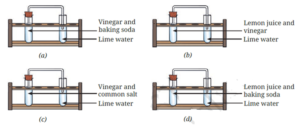
Question 10. The experiments shown in Fig. a, b, c, and d were performed. Find out in which case(s) did lime water turn milky and why?
Lime water turned milky in the cases (a) and (d) where vinegar or lemon juice was mixed with baking soda, because carbon dioxide gas was formed which reacts with
lime water to form a white precipitate (calcium carbonate).
The chemical reaction can be represented as follows
Vinegar or lemon juice + Baking soda -> Carbon dioxide + Other substances
Key Features of Kitabcd Exam Master :
|
Click on below links to get PDF from store
PDF : Chapter-5-Changes Around Us: Physical and Chemical– Notes
PDF : Chapter-5-Changes Around Us: Physical and Chemical– Exam Master
Main Page : NCERT-Class-7-Science (Curiosity) – All chapters notes, solutions, videos, test, pdf.
Next Chapter : Chapter-4- The World of Metals and Non-metals – Online Solutions
Next Chapter : Chapter-6- Adolescence: A Stage of Growth and Change – Online Solutions
We reply to valid query.